Rheology of Glass and Jamming
— Uncovering the Physical Commonality Between Mayonnaise and Glass —
(Collaborative research with the group of Prof. Masashi Ikeda, The University
of Tokyo)
We have demonstrated that the unusual viscoelastic properties observed
in “soft jammed solids,” such as mayonnaise and shaving foam, are deeply related to characteristic
vibrational modes found in glasses and amorphous solids—namely, quasi-localized
vibrations known as the boson peak.
Soft jammed solids are materials formed by densely packed, soft particles
arranged in a disordered manner, exhibiting properties intermediate between
liquids and solids. Although they are widely used in industrial products
and are abundant in everyday life, their fundamental mechanical propertieshave not been fully understood. In particular, the phenomenon of anomalously large viscous dissipationunder slowly applied external forces has long remained unexplained within
conventional viscoelastic theories.
To address this problem, we performed viscoelastic measurements of dense
emulsions using microrheology. By employing optical techniques, we precisely
measured the thermal fluctuations of embedded probe particles with high
spatiotemporal resolution. This approach enabled us to quantitatively capture
the anomalous viscoelastic response of soft jammed solids with unprecedented
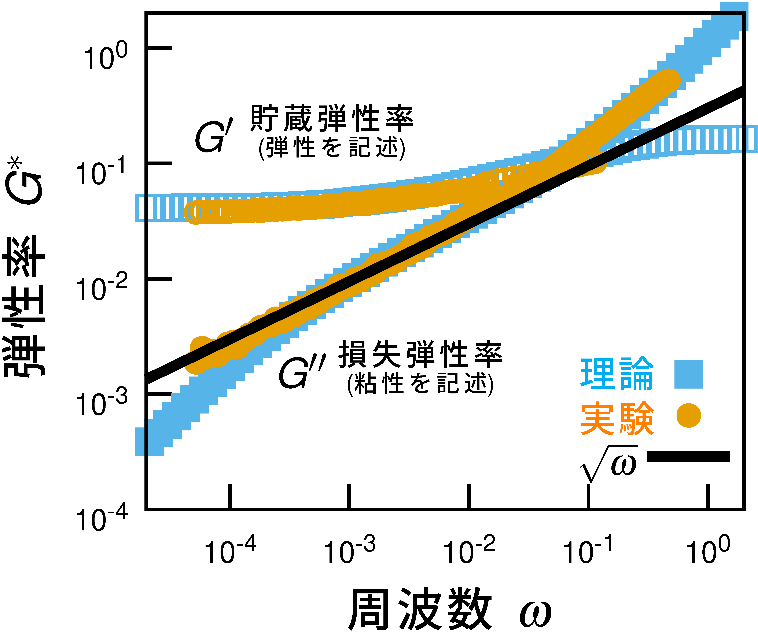
precision (Fig. 1).
A key finding is the clear scaling law in which theviscous loss increases proportionally to the square root of frequency (∝
ω¹ᐟ²). Such non-standard frequency dependence does not appear in ordinary viscoelastic
materials and is considered a hallmark of disordered structures.
Through detailed theoretical analysis of the experimental data, we found
that the origin of this phenomenon lies in dissipative vibrational modes
associated with the random contact network between particles—corresponding
to the boson peak. In glasses, vibrational modes propagating through disordered
contact networks include quasi-localized and non-elastic components that lead to energy dissipation. When subjected to an external oscillatory
force, these modes respond in a way that prevents energy storage and instead
promotes dissipation.
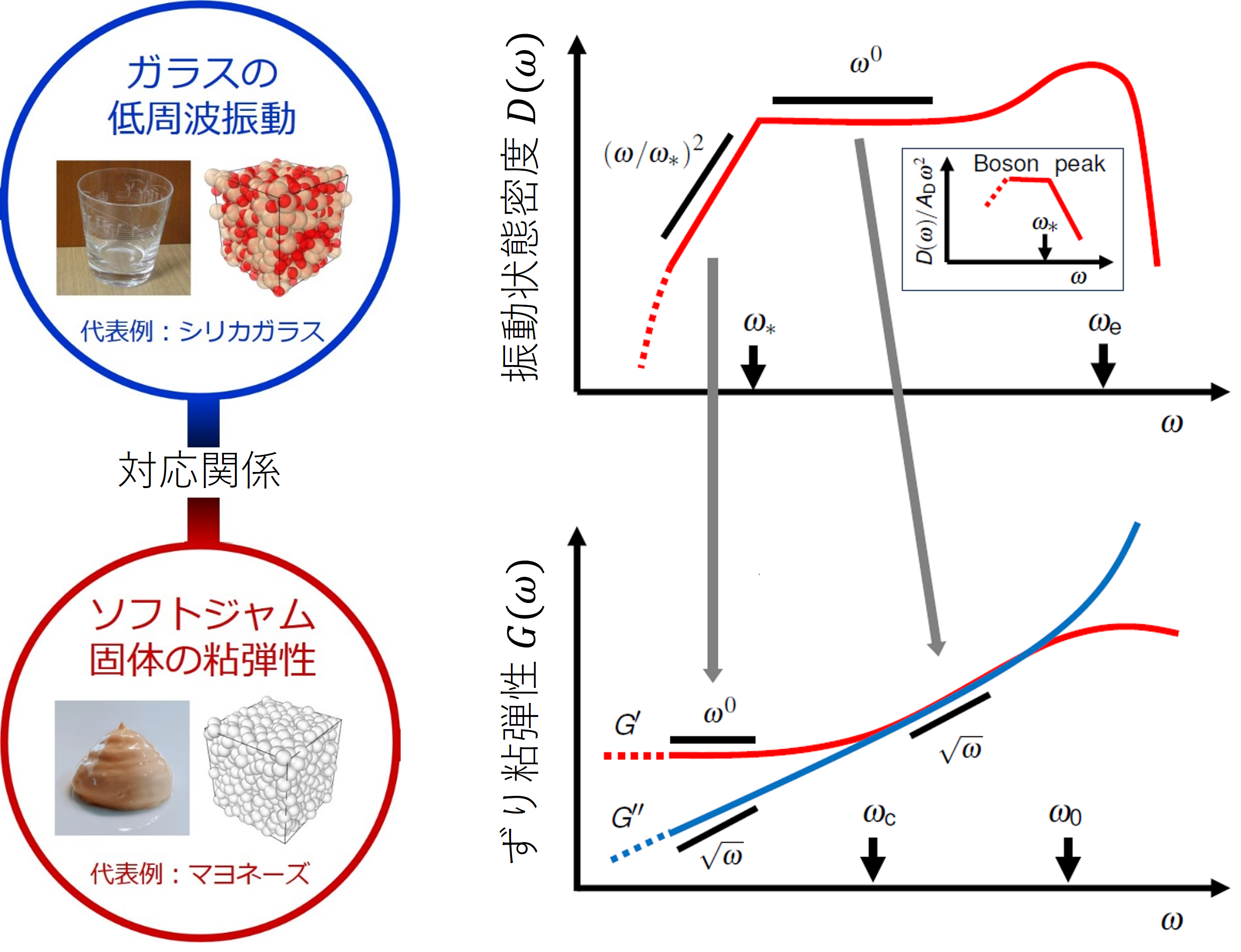
In soft jammed solids, anomalous relaxation modes emerge that correspond
to these glassy vibrational modes (Fig. 2), and they quantitatively reproduce
the observed ω¹ᐟ² scaling (Fig. 1).
This relationship is schematically illustrated in Fig. 2 of our study.
Understanding how structural disorder within soft jammed solids gives rise
to such anomalous relaxation modes—and how these modes dissipate energy
in response to external forces—is both an intriguing and challenging problem.
By clarifying the connection between glasses and soft jammed solids, our
work provides an intuitive framework for understanding the mechanism that
bridges macroscopic forcing and microscopic structural fluctuations through
energy dissipation.
This study represents one of the first demonstrations that soft matter
systems such as “softly jammed materials” and amorphous solids such as
disordered glasses can be described within a unified framework of condensed
matter physics. These findings are expected to provide new insights into
the physics of nonequilibrium materials and to guide the design of next-generation
materials.
This work has been published in Nature Physics.
DOI: 10.1038/s41567-024-02722-7
Non-equilibrium Rheology of Cells
Development of a Method to Directly Measure Nonequilibrium Rheology Inside Living Cells
We have successfully developed a method to measure the rheological properties
of the interior of living cells—namely stiffness, viscosity, and the magnitude of forces generated by the cell—which had long been considered technically inaccessible.
Rheology describes how materials flow and deform, and it is an essential
property for evaluating the performance and durability of industrial products
and advanced technologies. Similarly, to properly understand the function
and state of cells—the fundamental units of living organisms—it is highly
desirable to measure their rheological properties. However, cells are extremely
small (approximately 10 micrometers in diameter), and their interiors are
continuously driven by active processes powered by molecular-scale motors.
Accurately measuring material properties in such a small and dynamically
active environment has therefore been a major challenge. Until now, most approaches relied on probing the exterior of cells—effectively
“scratching” the surface—to infer properties near the membrane, making
it impossible to directly access the true internal state of the cell.
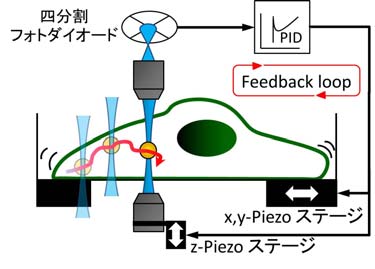
To overcome this limitation, we developed a method in which microscopic probe particles are introduced into living cells, and their
fluctuations and responses to externally applied forces are measuredwith high precision. Inside cells, vigorous motion known as cytoplasmic
streaming is constantly present. By employing feedback control, we were
able to compensate for this motion in real time and track the position
of the probe particles with sub-nanometer accuracy (Fig. 3).
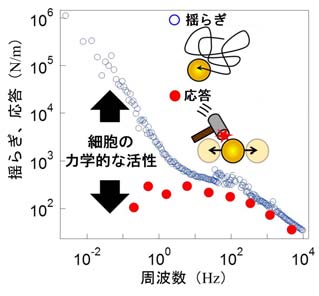
It is important to note that, in living cells, probe particles are actively
driven by the cell itself. Therefore, large fluctuations of the particles
do not necessarily indicate that the cell is soft or fluid-like (this interpretation
is only valid in dead cells). To address this, we measured the response
of the particles to controlled forces applied by laser trapping, allowing us to accurately extract the intrinsic rheological properties
of the cellular interior. Furthermore, by simultaneously measuring both the fluctuations and the
response of the particles, we were able to quantify how much force the
cell generates internally—effectively how “active” or “energetic” the cell
is—in terms of the violation of the fluctuation–dissipation theorem (Fig.
4).
The measurement technique we have developed thus provides the first direct
evaluation of the physical properties inside living cells. It is expected
to make significant contributions across a wide range of fields, including
physics, cell biology, and medicine. For further details, please refer
to ourreview articles and original publications.
-
Figure 3
The positions of a probe particle that vigorously flows insice a cell are traced by the multiple feedback via the laser and microscope stage.
-
Figure 4
A violation of the fluctuation-dissipation theorem serves as an indicator of cellular vitality (i.e., metabolic activity).
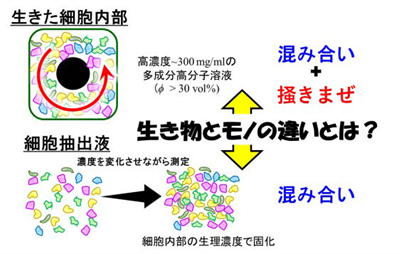
Cytoplasm as Active Glass
Crowding and “Stirring” Generate the Flexibility of Living Cells
We compared the viscoelastic properties (stiffness and viscosity) of living
cells with those of cell extracts, in which the cellular contents are removed
from their native context, in order to identify the origin of their differences.
Our results revealed that the combination of intracellular crowding and the stirring activity of
motor proteinsplays a decisive role (Fig. 5).
Cells can dynamically regulate their mechanical properties—such as stiffness
and viscosity—according to functional demands. In contrast, for inanimate
materials such as glasses or gels, altering their properties generally
requires restructuring the material itself. Living cells, however, can
modulate their properties in a far more flexible and dynamic manner. This
remarkable “flexibility” arises from the crowded intracellular environment
and the active stirring driven by motor proteins. If these factors strongly
influence the fluidity and viscosity of the material, then cells may indeed
possess fundamentally “lifelike” properties that distinguish them from
ordinary matter.
Without Crowding and Stirring, Cells Solidify
We first investigated cell extracts—prepared by disrupting the cell membrane and isolating the intracellular
contents—in which the stirring activity of motor proteins is effectively
removed. By varying the concentration of the contents, we measured their
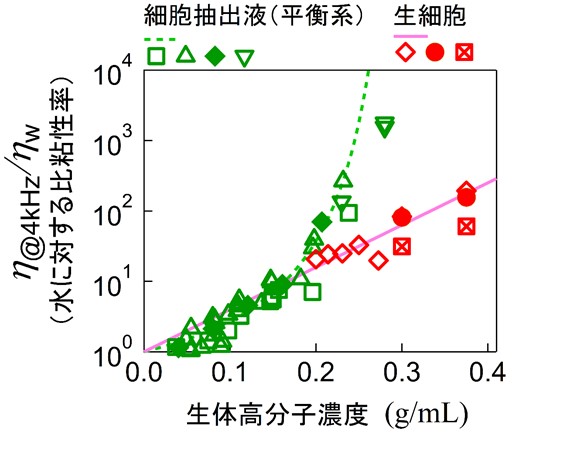
mechanical properties under these conditions. We found that evena modest increase in concentration led to a dramatic rise in viscosity, eventually causing the entire extract to solidify (Fig.6).
Remarkably, this phenomenon was observed across a wide range of cell types,
including human cells, bacteria, egg cells, and tissue cells. Furthermore,
solidification began at concentrations lower than those typically found
inside living cells (approximately 300 mg/mL). These results suggest that,in the absence of active stirring, cellular contents would readily become
solid-like. Such solidification would prevent essential processes such as molecular
synthesis and transport, ultimately impairing cellular function.
Why Do Living Cells Not Solidify?
To address this question, we measured the viscoelastic properties of living cellswhile similarly varying the intracellular concentration. Surprisingly,
despite having comparable concentrations, living cells maintained fluidity. Moreover, the manner in which viscosity changed with concentration was
entirely different from that observed in cell extracts. Detailed analysis
revealed that this difference originates from the cell’s intrinsic ability to actively “stir” its interior. In other words, cells prevent crowding-induced solidification through
the activity of motor proteins.
Until recently, intracellular crowding and active stirring had not been
central considerations in understanding cellular mechanics and function.
However, our findings suggest that these factors are crucial for cellular
flexibility, responsiveness, and potentially even cellular health. The mechanical properties of cells and tissues are known to influence
a wide range of physiological and pathological processes, including cancer progression, development, reproduction, and stem cell differentiation. Our results represent an important step toward a deeper understanding
of such phenomena and may open new avenues for applications across diverse
fields. For further details, please refer to the original publication.
-
Figure 5
Living cell and cellular extract
-
Figure 6
Concentration dependency of viscosity: living cell and cellular extract
Dynamics of Non-Gaussian Fluctuation
— Constructing a New Theoretical Framework for Statistics Unique to Nonequilibrium Systems —
Background: Non-Gaussian Fluctuations in Nonequilibrium Systems
In physical systems at equilibrium, fluctuations of observables are generally
expected to follow a Gaussian (normal) distribution, as guaranteed by the
central limit theorem. In particular, at the mesoscale—intermediate between
microscopic and macroscopic regimes—where systems can often be approximated
as homogeneous continua, measured quantities should exhibit Gaussian statistics.
However, in real nonequilibrium systems, this expectation is frequently
violated, and clearly non-Gaussian fluctuations are observed. Representative
examples include turbulence, glassy and jammed systems, intracellular force
generation and molecular transport, and suspensions of motile microorganisms
(active matter). By elucidating the origin of such non-Gaussian distributions, we can gain deeper insight into the physical properties and dynamics of nonequilibrium
systems through the shape of the distributions and their temporal evolution.
Construction of a New Non-Gaussian Limiting Distribution
In conventional statistical mechanics, when many independent contributions
with finite variance are superposed, the central limit theorem ensures
convergence to a Gaussian distribution. In contrast, when the system is
dominated by interactions with heavy-tailed distributions whose variance
diverges, the resulting distribution converges to a non-Gaussian stable
distribution known as a Lévy distribution. Such statistical limits naturally
arise in systems governed by long-range, power-law interactions, which
are ubiquitous in nature. For example, gravitational interactions between
stars in space, electrostatic forces in plasmas, and hydrodynamic interactions
among motile microorganisms all decay as the inverse square of distance.
Near each interaction source (e.g., particles, charges, or microorganisms),
the interaction strength diverges, leading to divergent variance when measured
at a mathematical point. In practice, however, measurements are always
performed over finite regions, and the variance remains finite.
This raises the following question: when a large number of interaction
sources are randomly distributed in three-dimensional space, what statistical
distribution emerges from the superposition of their interactions? If the
singular nature of individual interactions dominates, a Lévy distribution
is expected; if finite-size effects dominate, a Gaussian distribution should
arise. While the Gaussian approximation is often valid for equilibrium
systems, nonequilibrium systems frequently exhibit distributions that are
neither Gaussian nor Lévy.
To address this, we derived a new analytical expression for the limiting
distribution of fluctuations arising from interactions generated by randomly
distributed sources in three-dimensional space (e.g., motile microorganisms)
(see original paper).

The characteristic function (Fourier transform) of this distribution is
parameterized by:
• the characteristic system size R,
• the concentration of interaction sources c, and
• the interaction strength \gamma.
This formulation defines a new family of distributions that continuously
interpolates between Gaussian and Lévy distributions. Although we present
the explicit form for three dimensions here, a notable feature is that
the properties of the distribution depend on the spatial dimensionality.
Connection to Real Nonequilibrium Fluctuations and Outlook
This newly derived non-Gaussian distribution has the potential to quantitatively
describe fluctuations observed in a wide range of nonequilibrium systems,
including:
1. suspensions of motile microorganisms (active matter),
2. actin–myosin gels,
3. glassy soft matter, and
4. systems exhibiting turbulence or jamming.
We are currently combining experiments, theoretical analysis, and numerical
simulations to verify that this distribution accurately reproduces fluctuations
observed in real systems. For case (1), experimental validation has already
been completed and reported in our original publication. Furthermore, this
theoretical framework can be extended to incorporate the temporal dynamics
of the interaction sources themselves (e.g., microorganisms or molecular
motors), providing a pathway toward a more realistic and comprehensive
understanding of nonequilibrium dynamics.
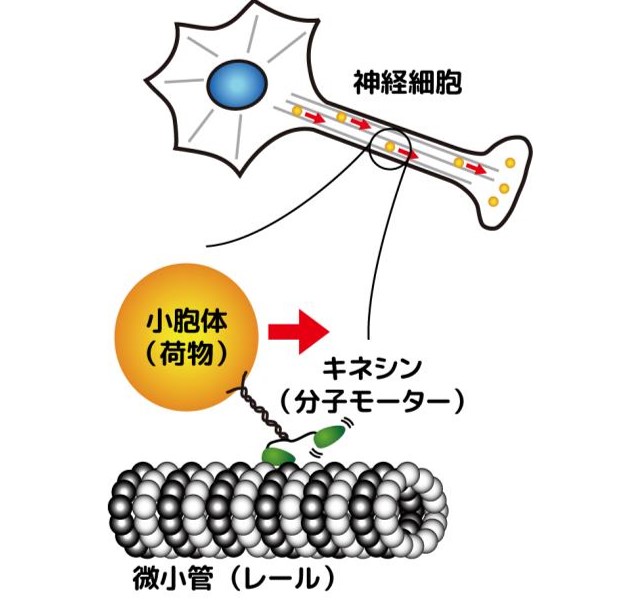
Fluctuations and Energetics of Biomolecular Machines
Collaborative research with Dr. Takayuki Ariga (Osaka University)
Using an optical tweezers system equipped with high-speed feedback control,
we have experimentally quantified the energy input and output at the single-molecule
level for the walking biomolecular motor protein kinesin (Fig.7). Through
mathematical modeling and theoretical analysis, we found that a large fraction
of the chemical energy supplied to kinesin is not used for cargo transport
but is instead dissipated as heat within the molecule.
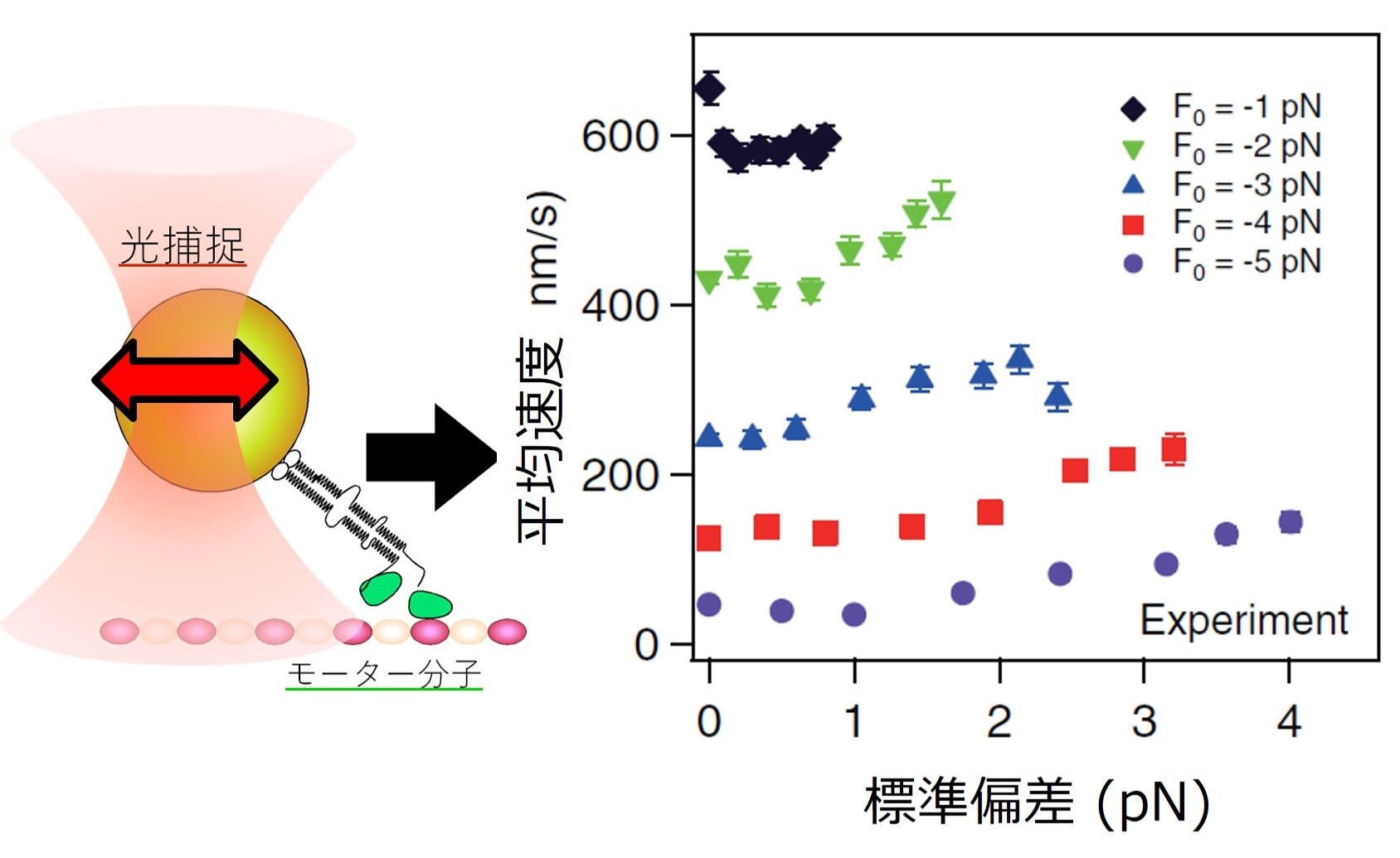
Furthermore, we discovered that kinesin can be accelerated by applying
artificially fluctuating forces that mimic the intracellular environment
(Fig.8). Notably, this acceleration becomes more pronounced under high
load conditions, suggesting that kinesin may be adapted to operate efficiently
in crowded and highly viscous cellular environments. These findings imply
that the non-thermal fluctuations present inside cells are not merely noise,
but may be actively utilized to enhance physiological functions (see original paper).
-
Figure 7
Molecular moter, kinesin, delivering a cargo.
-
FIgure 8
Increased velocity of kinesin in response to the fluctuating optical trapping force.
Nonequilibrium Mechanics Using an Exchange Chamber
Probing the Effects of Metabolic Activity on Intracellular Mechanical Environments
Inside living cells, the mechanical environment is shaped by the dynamics of biomolecules that consume energy carriers such as ATP. Recent studies suggest that,
although the intracellular environment is highly crowded and tends toward
a glass-like state, metabolic activity enables it to maintain fluidity. This raises key questions:
• How does metabolic activity influence the dynamic mechanical environment
inside cells?
• What are the molecular origins of intracellular fluidity?
Directly addressing these questions in living cells is challenging, because
cells exhibit feedback responses that maintain homeostasis. Attempts to externally manipulate metabolic or mechanical conditions
often trigger cellular responses, obscuring the underlying mechanisms.
A New Approach Using Cell Extracts and an Exchange Chamber
To overcome this limitation, we have developed a novel experimental system:
• Using cell extracts (intracellular contents isolated from cells), into
which metabolic activity can be artificially introduced.
• Allowing independent control of molecular concentration and metabolic activity.
However, in conventional closed systems, metabolic activity cannot be sustained over long periodsdue to depletion of active components and accumulation of waste products.
To address this, we developed an exchange chamber with the following features:
• Continuous supply of active molecules (e.g., ATP) through a semipermeable
membrane
• Simultaneous removal of metabolic byproducts
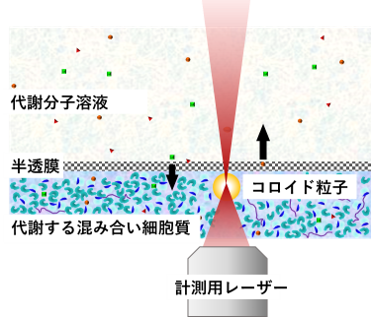
This setup enables long-term microrheology measurements under sustained metabolic activity (Fig. 9). Using this system, we can directly investigate the effects of
metabolism on intracellular mechanical properties without interference
from cellular feedback mechanisms.
-
FIgure 9
Micro-rheology measurement of cellular extract while sustaining metabolic activity.
-
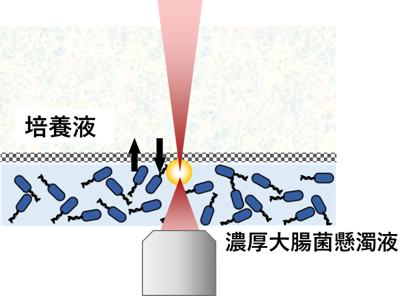
Figure 10
Micro-rheology measurement of dense suspension of E.coli.
Bacterial Suspensions as a Model Nonequilibrium System
As a model system mimicking the nonequilibrium intracellular environment,
we also study dense suspensions of Escherichia coli (Fig.10).
E. coli bacteria propel themselves by rotating flagella, driven by molecular
motors that are not directly observable under a microscope. By treating
the bacteria themselves as “visible motors,” we can effectively visualize
the stirring effects generated by otherwise invisible molecular processes
inside cells. At high concentrations, interactions among bacteria give
rise to collective swirling flows known as bacterial turbulence. Such systems belong to a broader class known as active matter, which
consists of self-driven particles that consume energy to generate motion.
Active matter has become a major topic of research in both physics and
biology.
Long-Term, Three-Dimensional Observation of Active Matter
Most previous studies of active matter have been limited to quasi-two-dimensional
systems, where motion can be observed but is short-lived due to depletion of energy sources. However, mechanical measurements require three-dimensional systems, which
have been difficult to realize. To overcome this, we constructed a three-dimensional active matter system using the exchange chamber. By continuously supplying nutrients and removing waste products through
a semipermeable membrane, we achieved stable, long-term self-propelled
motion of bacteria. Although three-dimensional observation is more challenging,
we have confirmed the presence of sustained bacterial turbulence in this system (Movie 1,2).
-
Movie 1
Long-lasting bacterila turbulence.
-
Movie 2
Velocity field of the bacterial turbulence (Movie 1).
Transition to an Active Glass State
At even higher bacterial concentrations, collective motion (swimming and
turbulence) gradually disappears, and rearrangements between bacteria become
suppressed. This state is known as an active glass, a quasi-arrested state in which energy is continuously supplied (through flagellar rotation),
yet motion becomes localized and structural rearrangements are effectively
frozen. This active glass state provides a useful model for understanding
how mechanical properties emerge under conditions of high density, sustained
activity, and nonequilibrium dynamics—conditions analogous to those inside living cells.
By combining the exchange chamber with bacterial suspensions, we have established
a powerful experimental platform for microrheology in metabolically driven
nonequilibrium environments. This system enables quantitative investigation
of:
• how cells and biomolecules generate nonequilibrium dynamics, and
• how these dynamics influence mechanical properties such as viscoelasticity
and fluidity.
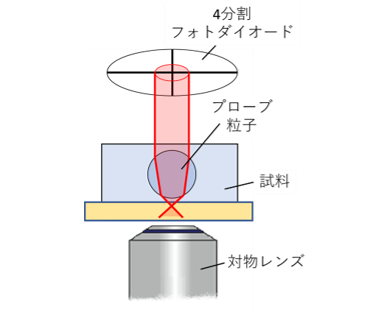
Microrheology Using Optical Lever Detection
To measure the mechanical properties of polymer networks such as gels using
microrheology, probe particles larger than the mesh size of the network
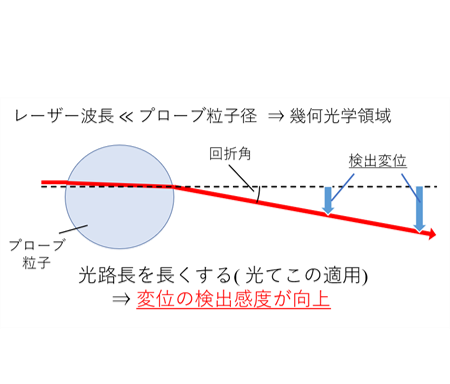
are required. However, in laser interferometry-based measurements, sensitivity
decreases when the probe size exceeds the optical wavelength (geometric
optics regime).
To address this issue, we have designed an optical system using an optical
lever, which maintains high sensitivity even for larger probe particles
(Fig.11,12). In this method, the transmitted laser beam is collimated by
the probe particle itself, effectively increasing the optical path length
and enhancing displacement sensitivity. In addition, by incorporating adaptive
optics, we aim to achieve high measurement sensitivity even in optically
heterogeneous samples.
-
Figure 11
Micro-rheology measurement using optical lever.
-
Figure 12
Mechanism of optical lever. By using the colloidal particles to collimate the laser light that has passed through them, increasing the optical path length improves the sensitivity of displacement detection.
Microrheology of Dense Colloidal Suspensions Under Shear
When macroscopic shear is applied to glassy systems, it is well known that
the viscosity exhibits nonlinear dependence on the applied stress. However,
the microscopic mechanisms underlying this nonlinear response remain unclear.
In our laboratory, we aim to elucidate these mechanisms by measuring mechanical
responses at the level of individual constituent particles under applied
macroscopic shear (Fig.13). In parallel, we also perform measurements of
microscopic mechanical responses to localized forces applied via optical
trapping, allowing direct comparison between macroscopic and microscopic
nonlinear dynamics.
We further complement these experiments with Brownian dynamics simulations
near the glass transition to deepen our understanding of these phenomena
(Movie 3).
-
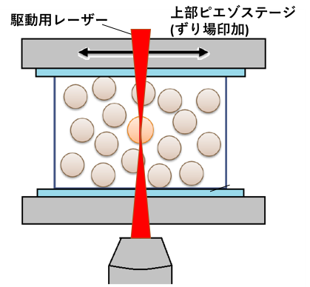
Figure 13
Micro-rheology under macroscopic shear field.
-
Movie 3
Brownian molecular dynamics simmulation near the glass transition.